Victor Ryzhov
Bioanalytical Mass Spectrometry
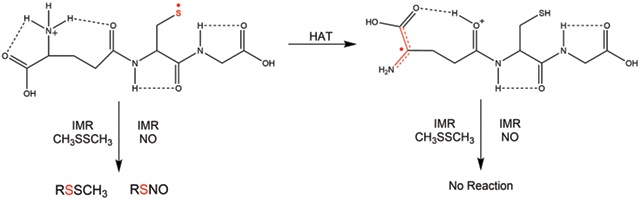
Due to the often transient nature of biological free radicals in solution, there has been an increased interest in gaining a more fundamental understanding of the structure and reactivity of radicals in small model systems in the gas phase. The main direction of our research has been to develop new methods for the study of radical ions in the gas phase using mass spectrometric (MS) techniques. In particular, we have employed ion–molecule reactions (IMR) for the study of radical ion reactivity. The model systems include amino acids with reactive amino acid side chains (cysteine, tyrosine, tryptophan), small peptides and DNA nucelobases where the radicals can be generated regiospecifically via various chemical means. Gas‐phase IMR of these radical ions shed light on their structure and provide convenient route to explore their intrinsic reactivity with the potential to study radical migration and radical‐induced damage. For example, we can monitor hydrogen atom transfer (HAT) in glutathione radical: the S‐based (thiyl), reactive radical is rearranging via HAT to a non‐reactive α‐carbon radical.
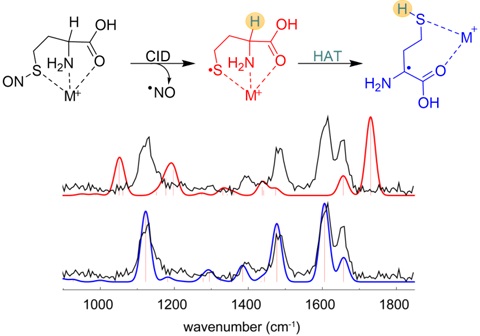
We also use ion spectroscopy (in IR and UV regions) and theoretical calculations to complement our gas‐phase reactivity studies. In the example below, isomeric thiyl and α‐carbon radicals of homocysteine are distinguished by ion IR spectroscopy.
Research Interests
- Bioanalytical mass spectrometry
- Gas‐phase ion reactivity and energetics
- Radical ions of biomolecules, gas‐phase catalysis using metal ion complexes
- HPLC and LC/MS assays for monitoring enzyme inhibition (collaboration with Prof. Hagen)
Representative Publications
- Fanizza, A.; Hoffman, J.; Gilbert, T. M.; Ryzhov, V. "Decarboxylation of Fatty Acids by Ternary Zinc Cationic Complexes Studied by Mass Spectrometry and Theoretical Calculations." Int. J. Mass Spectrom., 2024, 499, 117216.
- Parker, K.E., Bollis, N. and Ryzhov, V. "Ion-molecule reactions of mass-selected ions." Mass Spectrom Rev., 2024, 43(1), 47.
- King, R., Canty, A. J., Ariafard, A., O’Hair, R. A. J. and Ryzhov, V. "Catalytic Dehydrogenation of Liquid Organic Hydrogen Carrier Model Compounds by CpM+ (M = Fe, Co, Ni) in the Gas Phase." Organometallics, 2022, 41, 3823.
- Parker, K.; Mohr, A.; O'Hair, R. A. J.; Ryzhov, V. "Cracking and Dehydrogenation of Cyclohexane by [(phen)M(X)]+ (M = Ni, Pd, Pt; X = H, CH3) in the Gas Phase." Organometallics, 2021, 40(19), 3351.
- Parker, K.; Weragoda, G.; Pho, V.; Canty, A. J.; Polyzos, A.; O’Hair, R. A. J. and Ryzhov, V. "Gas-Phase Models for the Nickel- and Palladium-Catalyzed Deoxygenation of Fatty Acids." ChemCatChem, 2020, 122, 5476.
- Piacentino, E.; Parker, K.; Gilbert, T. M.; O’Hair, R. A. J. and Ryzhov, V. "Role of ligand in the selective production of hydrogen from formic acid catalysed by the mononuclear cationic zinc complexes [(L)Zn(H)]+ (L = tpy, phen, and bpy)." Chem. Eur. J., 2019, 25, 9959.
- Lesslie, M.; Yang, Y.; Canty, A. J.; Piacentino, E.; Berthias, F.; Maître, P.; Ryzhov, V. and O'Hair, R. A. J. "Ligand‐induced decarbonylation in diphosphine‐ligated palladium acetates [CH3CO2Pd((PR2)2CH2)]+ (R = Me and Ph)." Chem. Comm., 2018, 54, 346.
- Lesslie, M.; Lawler, J. T.; Dang, A.; Korn, J.A.; Bím, D.; Steinmetz, V.; Maître, P.; Tureček, F. and Ryzhov, V. "Cytosine Radical Cations: A Gas‐Phase Study Combining IRMPD Spectroscopy, UVPD Spectroscopy, Ion–Molecule Reactions, and Theoretical Calculations." ChemPhysChem, 2017, 18, 1293–1301. DOI: 10.1002/cphc.201700281
- Lesslie, M.; Lau, J. K.‐C.; Lawler, J. T.; Siu, K. W. M.; Oomens, J.; Berden, G.; Hopkinson, A. C. and Ryzhov, V. "Alkali‐Metal‐Ion‐Assisted Hydrogen Atom Transfer in the Homocysteine Radical." Chem. Eur. J., 2016, 22, 2243.
Contact

Professor and Chair
Faraday Hall 320
815‐753‐1181
ryzhov@niu.edu
Ryzhov Research Group
Educational Background
- Research Associate, University of Maryland, College Park, 1998–2000
- Ph.D., Case Western Reserve University, 1998
- M.S., Moscow State University, 1992