Yingwen Cheng
Materials Chemistry; Electrochemistry; Energy Chemistry
Our research lies at the interface between surface science, materials chemistry, electrochemistry and nanoscience. The goal is to develop advanced materials for sustainable energy storage and chemical conversion. This will be done through two focus areas: 1) fundamental understanding on the role of interfacial properties on the formation and phase transition of energy materials, and developing innovative approaches to synthesize materials with structure and property controlled across multiple length scale; 2) general reaction mechanisms for applications of these novel materials toward the next generation batteries, fuel cells and catalysts, and significantly improve the performance of the state‐of‐the‐art technology.
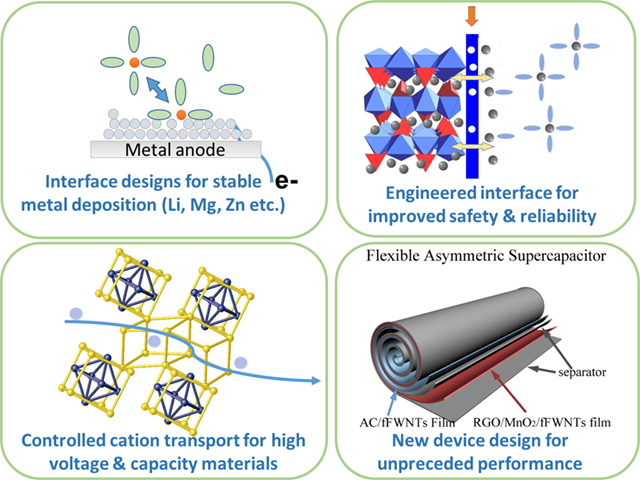
Efficient energy storage is critical for practical adoption of renewable energy and deployment of electric vehicles. Current technologies, such as lithium ion and lead–acid batteries, have intrinsic limitations and the need for the next‐generation batteries is urgent. Our research in this topic focuses on developing new technologies through fundamental understanding and controlling ion transport across electrochemical interfaces and within the host materials. The emphasis is to develop advanced battery materials and efficient artificial interfaces. Specific systems for the near future are 1) multi‐valent batteries, particularly those involve Mg metal; and 2) batteries with water based electrolytes, with water working as either the main solvent or an additive.
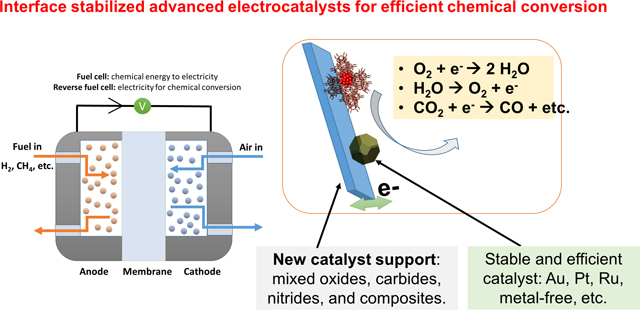
One of the key challenges in electrochemical/photochemical chemical conversion is the design of catalytic materials with earth‐abundant elements featuring high product selectivity and stability. We aim to develop novel nanostructured materials with desired electronic and geometric properties. We will apply these materials for catalytic reactions that are relevant to a cleaner and sustainable environment, particularly oxygen reduction reaction for fuel cells, oxygen evolution reaction for reverse fuel cells (electrolyzer), and electrochemical conversion of carbon dioxide and methane to energy dense liquid fuels. We are also interested in designing catalyst support materials with improved electrochemical durability especially during transient operation conditions. We will synthesis a series of nanomaterials with defined structure and compositions, including mixed oxides, carbides, nitrides and their composites. The fundamental binding properties of catalysts with these supports with respect to catalyst particle size, distribution, reactivity and stability will be systematically analyzed and optimized.
Representative Publications
Dahunsi, O.; Li, B.; Gao, S.; Lu, K.; Xia, F.; Xu, T.; Cheng, Y. "One-Step Synthesis of Na–Sn Alloy with Internal 3D Na15Sn4 Support for Fast and Stable Na Metal Batteries" ACS Applied Energy Materials, 2021, 5, 20-26
Lu, K.; Xia, F.; Li, B.; Liu, Y.; Abdul Razak I. ; Gao, S.; Kaelin, J.; Brown, D. E.; Cheng, Y.* "Synergistic Multisites Fe2Mo6S8 Electrocatalysts for Ambient Nitrogen Conversion to Ammonia" ACS Nano, 2021,15, 16887-16895
Xia, F.; Li, B.; Liu, Y.; Liu, Y.; Gao, S.; Lu, K.; Kaelin, J.; Wang, R.; Marks, T.; Cheng, Y. "Carbon Free and Noble Metal Free Ni2Mo6S8 Electrocatalyst for Selective Electrosynthesis of H2O2" Advanced Functional Materials, 2021, 2104716
Gao, S.; Li, B.; Lu, K.; Alabidun, S.; Xia, F.; Nickel, C.; Xu, T. and Cheng, Y. "Modulating MnO2 Interface with Flexible and Self-Adhering Alkylphosphonic Layers for High-Performance Zn-MnO2 Batteries" ACS Applied Materials & Interfaces, 2021, 13, 20, 23724-23731
Gao, S.; Xia, F.; Li, B.; Abdul Razak, I.; Liu, Y.; Lu, K.; Brown, D.; Wang, R. and Cheng, Y. "Synergistics of Fe3C and Fe on Mesoporous Fe–N–C Sulfur Host for Nearly Complete and Fast Lithium Polysulfide Conversion" ACS Applied Materials & Interfaces, 2021, 13, 15, 17791-17799
Lu, K.; Li, B.; Zhan, X.; Xia, F.; Dahunsi, O.; Gao, S.; Reed, D.; Sprenkle, V.; Li, G. and Cheng, Y. "Elastic NaxMoS2-Carbon-BASE Triple Interface Direct Robust Solid–Solid Interface for All-Solid-State Na–S Batteries" Nano Letters, 2020, 9, 6837-6844
Zhang, H.; Lu, K.; Li, B.; Liu, Y.; Su. Y.; Wang, R. and Cheng, Y. "Microfluidic, One-batch Synthesis of Pd Nanocrystals on N-doped Carbon in Surfactant-free Deep Eutectic Solvents for Formic Acid Electrochemical Oxidation" ACS Applied Materials & Interfaces, 2020, 12, 42704-42710
Lu, K.; Xu. H.; He, H.; Gao, S.; Li, X.; Zheng, C.; Xu, T. and Cheng, Y. "Modulating reactivity and stability of metallic Li via atomic doping" Journal of Materials Chemistry A, 2020, 8, 10363-10369
Lu, K.; Liu, Y.; Lin, F.; Cordova, I.; Gao, S.; Li, B.; Peng, B.; Xu, H.; Kaelin, J.; Coliz, D. ; Wang, C.; Shao, Y. and Cheng, Y. "LixNiO/Ni Heterostructure with Strong Basic Lattice Oxygen Enables Electrocatalytic Hydrogen Evolution with Pt-like Activity" Journal of the American Chemical Society, 2020, 142, 29, 12613-12619
Lu, K.; Liu, Y.; Chen, J.; Zhang, Z.; Cheng, Y. "Redox Catalytic and Quasi-Solid Sulfur Conversion for High Capacity Lean Lithium Sulfur Batteries" ACS Nano, 2019, 13, 12, 14540-14548
Lu, K.; Zhang, H.; Gao, S.; Ma, H.;* Chen, J. and Cheng, Y. "Manipulating Polysulfide Conversion with Strongly Coupled Fe3O4 and Nitrogen Doped Carbon for Stable and High Capacity Lithium-Sulfur Batteries" Advanced Functional Materials, 2018, 1807309

Adjunct and Visiting Faculty
La Tourette Hall 411
815‐753‐7033
ycheng@niu.edu
yingwencheng.org
Educational Background
PostDoc, Pacific Northwest National Laboratory, 2013–2017
Ph.D. (Chemistry), Duke University, 2013
B.S. (Chemistry) and B.Eng. (Chemical Engineering). Shandong University, 2008
Research Interests
Development of novel electrode materials and electrolytes for stronger and more reliable beyond lithium–ion batteries. Novel electrocatalysts for efficient conversion of electricity into chemicals and energy‐dense liquid fuels. Materials synthesis with structure‐properties controlled across multiple length scales. In‐situ and ex‐situ advanced characterization.