Research
Gas-Phase Catalysis with Mass Spectrometry
By removing solution-based complications such as ion-pairing and solvent shell interactions a deeper understanding of catalytic processes and intermediates formed can be found when studying these processes in the gas-phase. Mass spectrometry is a complete gas-phase laboratory that allows for high specificity through ion-molecule with the introduction of volatile neutrals into in house modified ion traps. Activation of catalytic species via collision induced dissociation leads to decarboxylation of carboxylic acid complexes.
[(L)M(OOCR)]+ à [(L)M(R)]+ + CO2
These types of decarboxylated ions have a wide-ranging set of applications for organometallic catalysis.
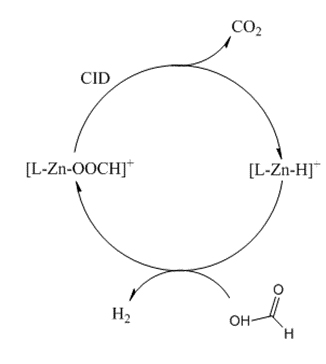
Hydrogen is a great fuel source for green energy and could lead to a reduction in dependence on fossil fuel products. Storing hydrogen chemically as formic acid improves the safety and stability of H2 during transportation and storage.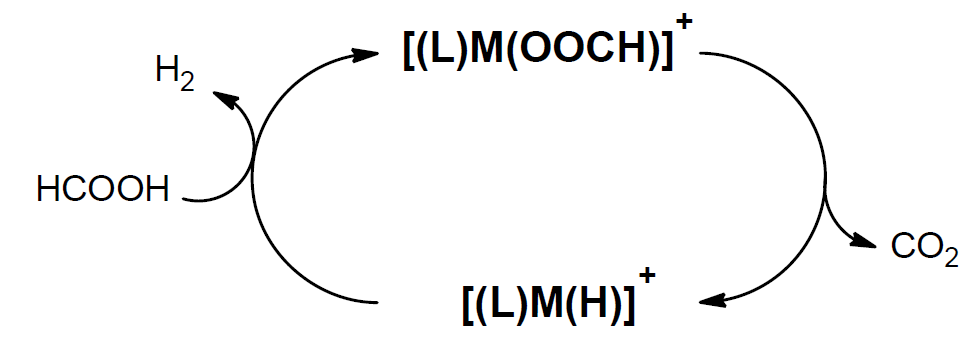
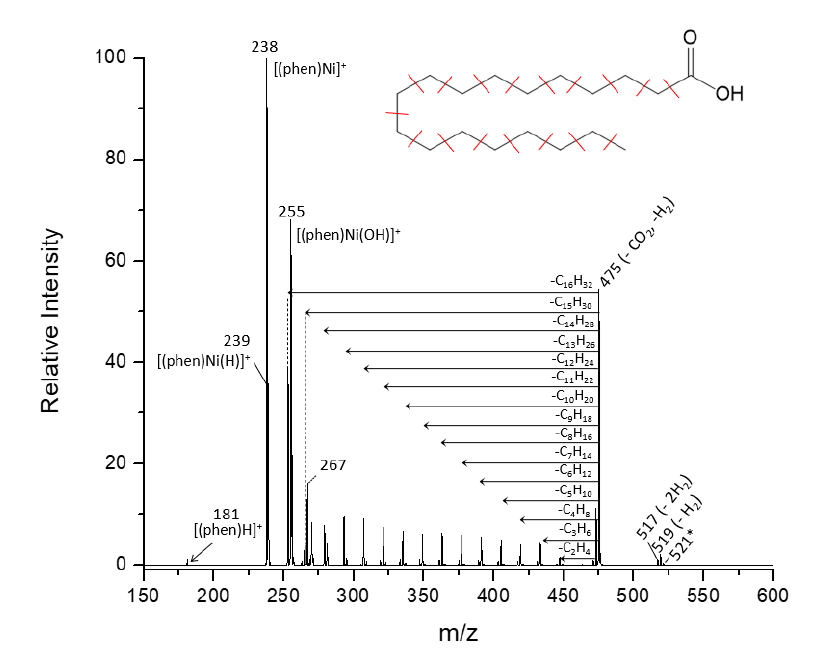
Chain walking reactions of group 10 metals (Ni, Pd, and Pt) can facilitate depolymerization and cracking of hydrocarbon chains like hexane and those found in fatty acids.
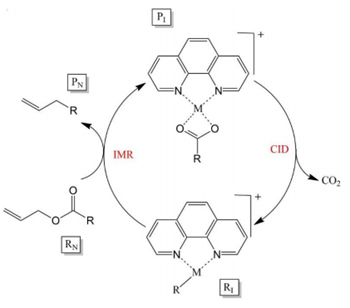
Formation of new bonds C-C and C-N bonds is a critical reaction in organic chemistry. Suzuki C-C coupling and Buchwald C-N coupling reactions both rely on metal catalysts. Studying these catalyst in the gas-phase allows for minimal amounts of metal used while screening for catalytic efficiency.
Bioanalytical Mass Spectrometry
Due to the often transient nature of biological free radicals in solution, there has been an increased interest in gaining a more fundamental understanding of the structure and reactivity of radicals in small model systems in the gas phase. The main direction of our research has been to develop new methods for the study of radical ions in the gas phase using mass spectrometric (MS) techniques. In particular, we have employed ion–molecule reactions (IMR) for the study of radical ion reactivity. The model systems include amino acids with reactive amino acid side chains (cysteine, tyrosine, tryptophan), small peptides, and DNA nucelobases, where the radicals can be generated regiospecifically via various chemical means. Gas‐phase IMR of these radical ions shed light on their structure and provide convenient route to explore their intrinsic reactivity with the potential to study radical migration and radical‐induced damage. For example, we can monitor hydrogen atom transfer (HAT) in glutathione radical: the S‐based (thiyl), reactive radical is rearranging via HAT to a non‐reactive α‐carbon radical.
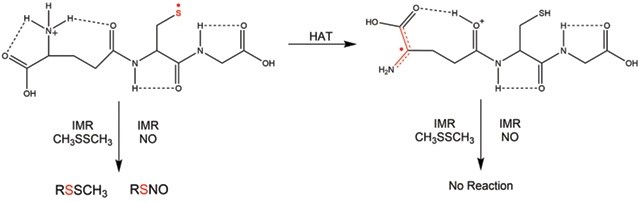
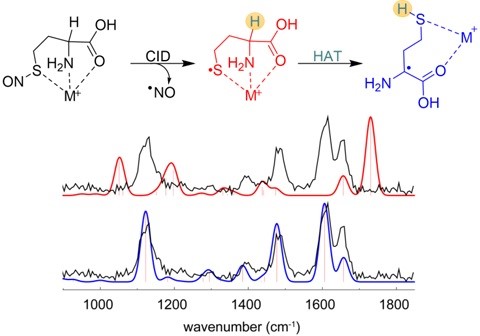
We also use ion spectroscopy (in IR and UV regions) and theoretical calculations to complement our gas‐phase reactivity studies. In the example below, isomeric thiyl and α‐carbon radicals of homocysteine are distinguished by ion IR spectroscopy.
Chromatography and Mass Spectrometry to Study Enzyme Inhibition
This research is focused on developing liquid chromatography-mass spectrometry (LC-MS) methods to study the efficacy of potential novel antibiotics. In collaboration with Prof. Horn and Prof. Hagen groups here at NIU, we target the non-mevalonate (MEP) pathway, which controls isoprenoid biosynthesis, as it is only present in bacteria and some plants and therefore extremely attractive for antibiotic development.
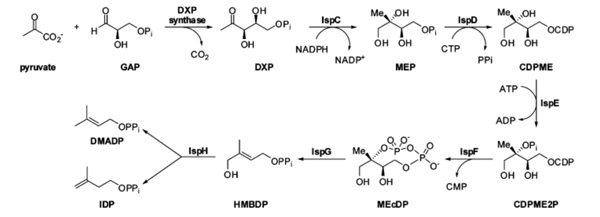
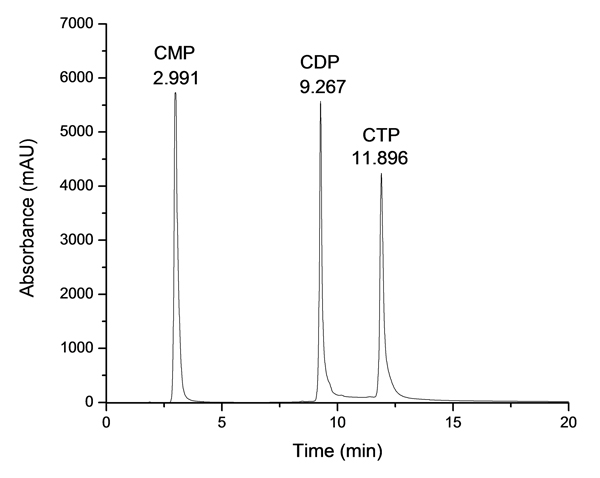
The MEP pathway primarily utilizes phosphate-containing groups in the biosynthesis of isoprenoid precursors. The extreme polarity of these species presents a challenge for chromatographic assay development, as conventional techniques such reverse phase cannot retain the analytes. We have found several other varieties of LC suitable to analyze phosphate containing groups, including anion-exchange (AX), ion-paired reversed-phase (IP-RP), and hydrophilic interaction (HILIC). Using these chromatographic techniques, coupled with mass spectrometry detection, we evaluate the effectiveness of potential inhibitors of the enzymes IspD, IspE, and IspF of the MEP pathway in two ways: time-based solution studies and cell growth inhibition studies.
Investigation of metal-ion catalysts in the gas phase
Our interest is focused on the development of new catalysts materials and in the study of their fundamental mechanism of action as well as their potential applications. It is known that precious metals-based complexes are able to catalyze C-C coupling reactions as well as participate in the catalytic cycle of hydrogen production from chemical hydrogen storage materials such as formic acid. However, these metals are not naturally abundant are expensive. For this reason, the exploration of less expensive materials sharing the same catalytic abilities is of fundamental importance for future applications of the processes on a larger scale.
Our laboratory is equipped with a custom modified mass spectrometer which allows for the study of ion-molecule reactions (IMRs) in the gas phase. The combination of gas-phase experiments, IMRs, and MSn ability permits the scan of the catalytic aptitude of many candidate molecules but also allows for the evaluation of the kinetics properties of the systems. Mass spectrometry experiments combined with computational studies aids in a complete evaluation of the catalytic systems.
Below is an example of the catalytic cycle to produce hydrogen from formic acid when a Zn based catalyst is used. The complex ion is constituted by the metal coordinating a ligand (L), which can be varied to tune the complex properties, and a formate ion. The cycle can be divided into two steps, both conducted inside the MS ion trap. The first reaction, slightly endothermic, results in the decarboxylation of the metal complex, and the second reaction is between the hydride form of the complex ion, produced in the decarboxylation step, and formic acid; it is spontaneous and it yields H2 as well as restoring the zinc catalyst.